This article provides an overview of Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) and Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS): A Beginner’s Guide for Mental Health Providers and Allied Professionals
Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) is a clinical condition characterized by the sudden onset of severe neuropsychiatric symptoms in children, most notably obsessive-compulsive behaviors (OCD) and/or severe food restriction, often accompanied by a range of other cognitive, emotional, and behavioral changes. Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) is a subset of PANS. It refers to a specific autoimmune condition in children in which a streptococcal infection triggers a sudden and severe onset of OCD symptoms and/or tics in a previously healthy child. These disorders are characterized by many of the same neurological and psychological symptoms (PANDAS Network, 2025). In both PANS and PANDAS, the onset of symptoms is considered abrupt, usually occurring within 1 to 3 days, and sometimes described as overnight. Parents often report that the child seemed fine one day and suddenly showed severe symptoms on the following day. Rapid onset symptoms of these disorders range from obsessions-compulsions to insomnia to mood instability (Rea et al., 2021). As such, these conditions are difficult to accurately diagnose due to the complicated nature of linking streptococcal infection to the onset of psychiatric symptoms (Rea et al., 2021). A critical consequence of misdiagnosis is that individuals affected by these rare conditions do not receive appropriate care in a timely manner. Encouragingly, treated early and in a timely fashion, PANS/PANDAS can remit entirely (PANDAS Network, 2025).
The adoption of a multidisciplinary collaborative approach has the potential to improve not only diagnosis but also short- and long-term treatment outcomes for clients (Aspire, 2025).
While mental health professionals are not able to diagnose PANS/PANDAS, they can play an important role in a multidisciplinary approach. For instance, mental health clinicians can help identify clients exhibiting symptoms of PANS/PANDAS and refer them to professionals with appropriate expertise. These professionals may include pediatricians, child psychiatrists, and neurologists, among others. Further, mental health clinicians can (a) collaborate with other professionals to ensure that any co-occurring psychological symptoms or conditions are addressed and (b) support families in understanding and managing these conditions.
This article serves as a beginner’s guide to PANS/PANDAS for mental health professionals and those in related disciplines. Topics discussed include the definitions, etiologies, consequences (e.g., individual- and family-level), and evidence-based practices (e.g., assessments and interventions) related to PANS and PANDAS. These topics will be illustrated with a clinical vignette and further reinforced with key takeaway points. Improved recognition, assessment, diagnosis, treatment, and management offer the opportunity for positive outcomes for clients and their families.
PANS and PANDAS
Although the lifetime prevalence remains unclear, approximately 1 in 200 American children are estimated to experience PANS and/or PANDAS. This estimated prevalence is roughly comparable to that of conditions such as pediatric cancer and pediatric diabetes (PANDAS Network, 2025). PANS and PANDAS usually affect children ages 4 to 13 (Thienemann et al., 2016), with boys more likely to be diagnosed than girls. Both conditions have been linked to abnormal autoimmune responses to bacterial exposure (Thienemann et al., 2016). The primary focus has been responses to streptococcal infections; however, a variety of suspected infections have been associated with PANS. These suspected infections are mostly upper respiratory infections, including pharyngitis, bronchitis, and rhinosinusitis (Piras et al., 2020).
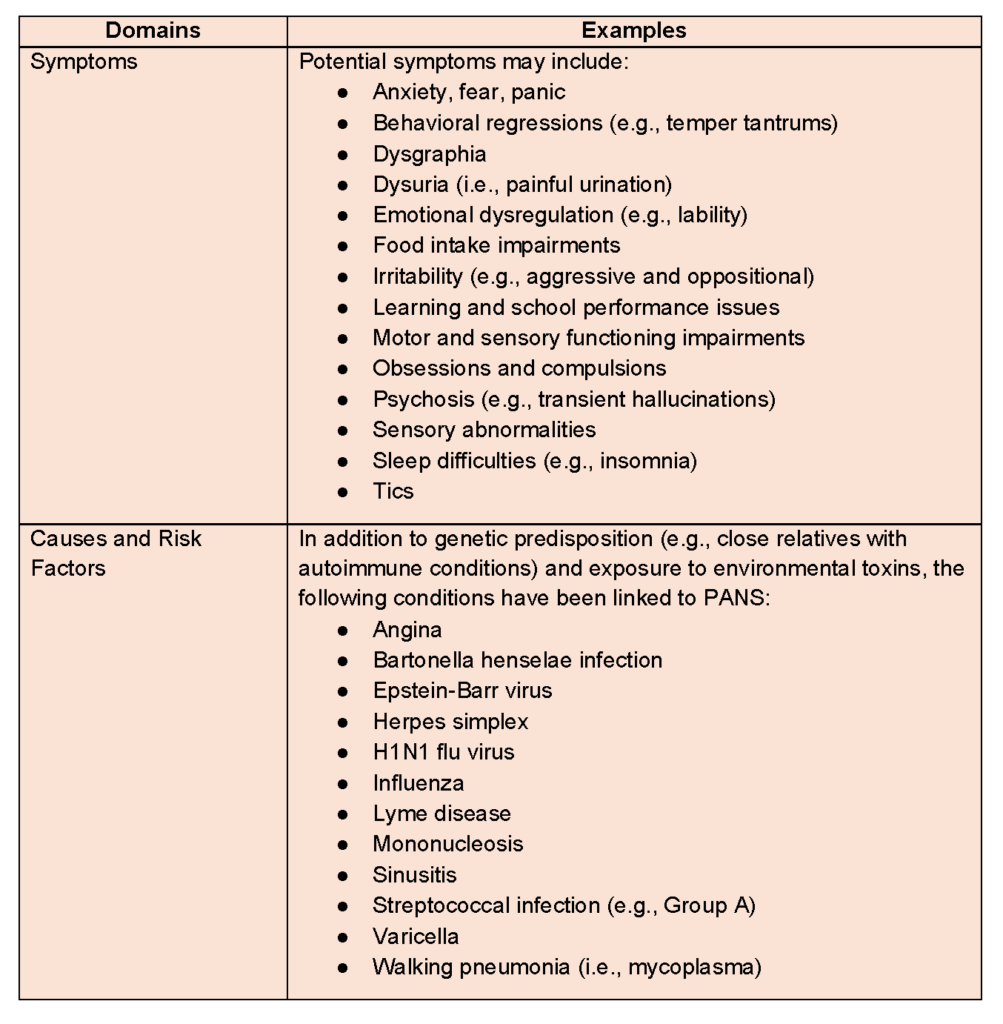
Table 1 highlights the overlapping symptoms and consequences of PANS and PANDAS (Bejerot et al., 2021; LaRusso & Abadía-Barrero, 2024).
Table 1. Overlapping Symptoms and Consequences of PANS and PANDAS.
PANS
PANS is a relatively new clinical condition that remains misunderstood, controversial, and underdiagnosed. A clinical diagnosis of PANS requires the sudden onset of (a) obsessive-compulsive symptoms or severe restriction in food intake and (b) at least two different neuropsychiatric symptoms not attributable to another condition (Biswas et al., 2022; Chan et al., 2022), such as Tourette’s syndrome (Baytunca et al., 2016). Although the etiology of this condition remains uncertain, many researchers believe PANS may be precipitated, at least in part, by exposure to pathogens, infections, or autoimmune diseases (Alqifari & Maxwell, 2023; Aman et al., 2022). In addition to these and other illnesses, the etiology of PANS is likely influenced by genetic and environmental risk factors (e.g., specific foods, toxins, and psychological distress; Arcilla & Singla, 2024; Baj et al., 2020; Calaprice et al., 2017).
Over the course of studying the etiology of PANS, the most frequently reported single microbe consistently associated with its onset was Group A hemolytic Streptococcus. Streptococcus is known to induce antibody production, including those antibodies that mimic basal ganglia epitopes, which are specific regions on an antigen that are recognized by the immune system (Piras et al., 2020). Thus, the theory of immune mechanisms in neurodevelopmental disorders is based on the mechanism of ‘molecular mimicry’ (Piras et al., 2020). The theory proposes that serum antibodies produced against non-infectious and infectious agents may cross the blood-brain barrier (BBB) and cross-react with antigens on healthy neuronal cells. This cross-reaction may then lead to dysregulation of basal ganglia function, producing a variety of neurological and psychiatric symptoms. (Piras et al., 2020). Normally, the BBB prevents circulating antibodies from entering the central nervous system, but in PANS, this barrier’s compromise is a key factor.
This immune response extends to neural circuits, with current evidence supporting the involvement of cortico-striato-thalamo-cortical (CSTC) circuits and basal ganglia inflammation in PANS/PANDAS. MRI and clinical studies have reported basal-ganglia abnormalities and antibody-mediated changes in these regions (Piras et al., 2020; Gagliano et al., 2024). Although direct functional-MRI connectivity studies are limited, findings from related autoimmune and obsessive-compulsive spectrum research suggest dysregulation within CSTC loops. Reports of inflammatory markers and antibody binding to striatal cholinergic interneurons further substantiate the neuroimmune hypothesis (Rea et al., 2021). While neurotransmitter imbalances, particularly dopaminergic and glutamatergic, are biologically plausible contributors (Xu et al., 2021), direct neurochemical evidence in PANS/PANDAS remains preliminary.
This “post-infection” immune activation also suggests the involvement of the human leukocyte antigen (HLA) system, which may aid the antibodies in crossing the BBB (Gagliano et al., 2024). The mechanisms by which the serum antibodies produced enter the brain to interfere with the functions of the basal ganglia are currently being elucidated. The scientific body of evidence for explaining “autoimmune PANS” is still in its infancy; however, there is currently substantial evidence supporting the inflammatory involvement of the basal ganglia and other neural networks (Gagliano et al., 2024).
Other microorganisms have been associated with the onset of PANS. The current list of additional microorganisms includes Mycoplasma pneumoniae, Borrelia burgdorferi, Staphylococcus aureus, Epstein–Barr virus, Influenza virus, Coxsackie virus, and Varicella-Zoster virus (Leonardi et al., 2024). Studies linking Tourette’s syndrome to Mycoplasma pneumoniae DNA in patients’ cerebrospinal fluid and to antibodies in patients’ blood to determine the association between the infection and tics were conducted in the early 2000s (Müller et al., 2000; Müller, 2004). A more recent Müller lab group study included 302 chronic tic disorder patients (CTD) (ages 3-16) in addition to their siblings with less severe tics or no tics at all. The findings of the study indicate that while there may not be a causal relationship between Mycoplasma pneumoniae infection and the development of tics (severe or not), there may be an indirect link via shared underlying immune mechanisms (Schnell et al., 2022).
There is also growing evidence of genetic risk factors within the pathogenic model of PANS. Children who inherit risk factors that lead to dysregulation of immune pathways have been known to exhibit PANS-like symptoms (Gagliano et al., 2024). Other risk factors, such as abnormally functioning neurosynaptic regulatory genes, could also be involved. Whole Genome Sequencing (WGS) and whole exome sequencing (WES) have now been used to pinpoint a small set of those genetic risk factors that may have a causal effect on PANS (Trifiletti et al., 2022).
Table 2 provides an overview of the symptoms (Baj et al., 2020; Bhattacharjya & Tona, 2016; Biswas et al., 2022; Baytunca et al., 2016; Silverman et al., 2019) and potential causes (Nadeau et al., 2015) of PANS. Studies of children with PANS have reported structural differences in the brain, including the amygdala, thalamus, basal ganglia, and deep gray matter (O’Hara, 2022). MRI research using atlas-based analysis has found that children with PANS showed significantly increased diffusivity across nearly all examined brain regions—particularly in the deep gray matter structures such as the thalamus, basal ganglia, and amygdala—while no significant differences in volume or cerebral blood flow were detected (Zheng et al., 2020). These microstructural differences, which were most pronounced in the basal ganglia and limbic circuits, align closely with the clinical presentation of obsessions, compulsions, emotional dysregulation, and sleep disturbance observed in PANS (Zheng et al., 2020). Inflammation has also been detected in the thalamus, amygdala, and basal ganglia (Rea et al., 2021). If not effectively treated, PANS can undermine a child’s short- and long-term well-being (Ringer, Benjaminson, & Bejnö, 2023), and may persist into adulthood (Frankovich et al., 2015; Schön, 2023).
Table 2. PANS: Symptoms and Causes.
PANDAS
Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) was initially reported in 1998 as a subtype of PANS by the National Institute of Mental Health. This condition is characterized by a sudden onset following Group A beta-hemolytic streptococcal (GABHS) oropharyngeal infections (Alexander et al., 2011). Diagnostic criteria include (a) the presence of obsessions, compulsions, and/or tics, (b) neuropsychiatric symptoms (e.g., anxiety and hyperactivity), and (c) a relatively young age of onset (between 3 to 12 years of age; Arcilla & Singla, 2024; Chang et al., 2015; Pandas Physicians Network, 2025; Thienemann et al., 2017). Estimates as high as 25% of children diagnosed with pediatric OCD and/or Tourette’s syndrome also have PANDAS (Gagliano et al, 2024).
The etiology and symptoms of PANS and PANDAS overlap. PANS cases may be temporarily associated with Group A Streptococcus (GABHS) infection, whereas PANDAS cases require a prior GABHS infection. Patients exhibiting acute-onset tics may be PANDAS cases, but not PANS cases. Abrupt onset of restrictive eating (with or without infection) may be a PANS criterion but not a PANDAS criterion. A PANS diagnosis requires at least two other abrupt-onset neuropsychiatric symptoms in addition to OCD or eating restrictions; however, PANDAS criteria do not require additional symptoms. One main overlap is the presence of an abrupt onset of OCD and previous GABHS infection (Leonardi et al., 2024).
PANDAS Typical Clinical Illustration
The following is an illustration of a typical presentation of a child with PANDAS.
An 8-year-old boy with no prior psychiatric history developed an abrupt onset of severe obsessive-compulsive symptoms within days of recovering from a streptococcal throat infection. The boy’s parents reported that the child began washing his hands repeatedly for hours, began restrictive eating, had difficulty sleeping, became unable to attend school due to separation anxiety, and displayed sudden mood lability and irritability. Additionally, there was a regression in academic functioning. The parents report that these symptoms appeared almost overnight, in contrast to the child’s previously stable personality.
Concerned about the abrupt behavioral changes, the boy’s parents consulted a mental health professional for evaluation and support. The clinician conducted an initial assessment and recognized that the sudden, severe onset of OCD and anxiety was not consistent with typical gradual-onset psychiatric disorders. Based on the clinical picture and recent history of streptococcal infection, the clinician referred the boy for further medical evaluation. The child underwent additional diagnostic testing and began a multidisciplinary treatment plan, which included medical management, PANDAS specialists, and supportive psychotherapy.
Following the multidisciplinary treatment plan, the child’s symptoms gradually improved over several weeks. The frequency and intensity of OCD behaviors diminished, and his food restriction and separation anxiety lessened, allowing him to return to school with minimal support. His mood had stabilized at school, and the boy was once again engaging in family activities and sleeping through the night without distress. Although occasional flare-ups occurred, the child demonstrated a return to prior functioning, consistent with a favorable recovery in PANDAS when proper interventions are implemented.
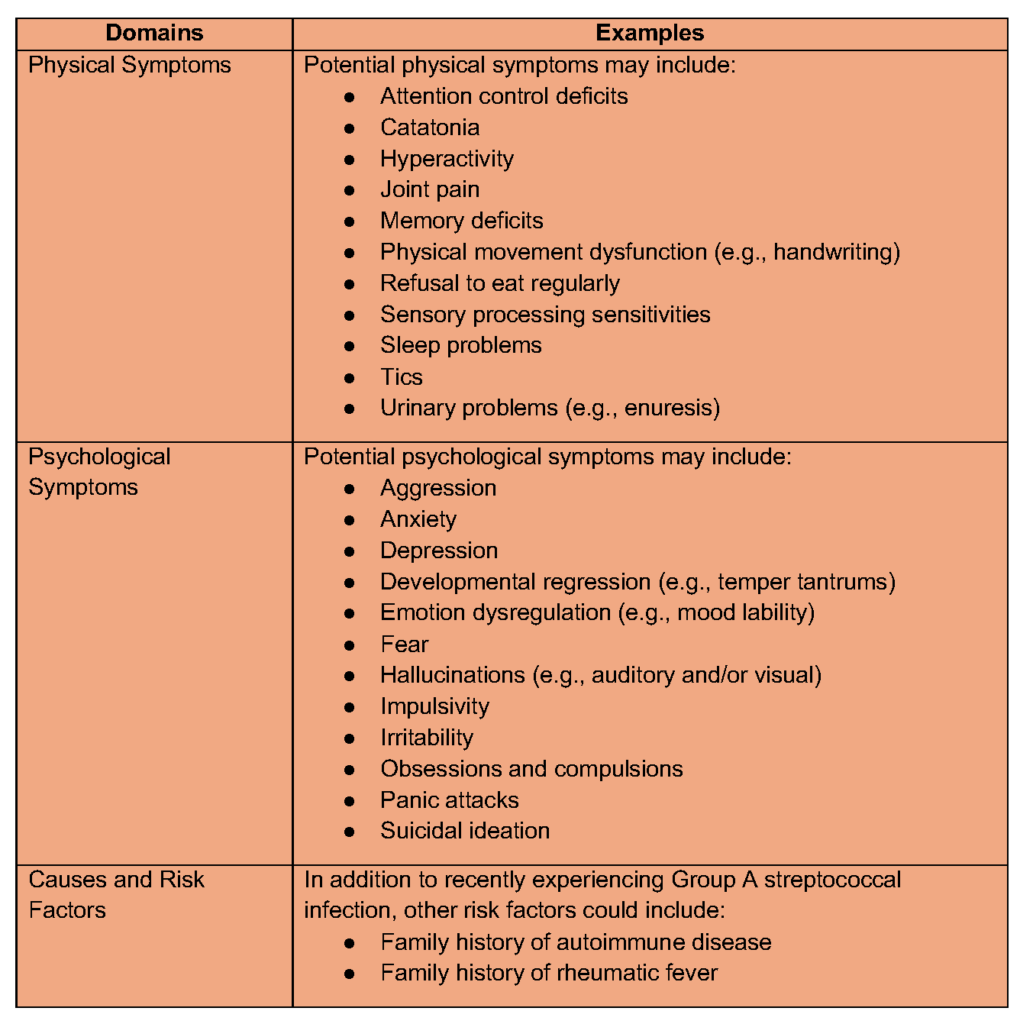
Table 3 provides an overview of the symptoms, as well as the potential causes and risk factors associated with PANDAS (Arcilla & Singla, 2024; Baj et al., 2020; Demchick et al., 2019; Sigra et al., 2018). These symptoms typically appear around a month after a streptococcal infection (Arcilla & Singla, 2024). The immune response may take time to affect the brain, meaning psychiatric symptoms could appear days, weeks, or a month after the infection, although the symptoms still emerge rapidly when they do appear. Although prevalence appears to be higher in males than in females, PANDAS remains a relatively rare condition (La Bella et al., 2023). Current theories suggest that PANDAS, like PANS, may result from an autoimmune response similar to what is observed in rheumatic fever (Blum et al., 2022). While the etiology remains unclear, microglia and the basal ganglia may play a role in the development of PANDAS (Baj et al., 2020). PANDAS patients have been found to have striatal and basal ganglia inflammation (Rea et al., 2021). Further, dysregulation of the immune system has been found in Tourette’s syndrome, including autoimmune-like activation of T-Cells and B-Cells, along with high levels of proinflammatory cytokines, including Interleukin-17A, Interleukin-6, Interleukin-12, and Tumor Necrosis Factor-α (TNF-α) (Leonardi et al., 2024). These, and other elevated bioactive anti-inflammatory markers such as basal ganglia antibodies, have been found in tic-disorder patients and OCD patients (Connery et al., 2018; Hsu et al., 2021). There is a clear need for research to identify clinical biomarkers of active inflammation in PANS/PANDAS (Connery et al., 2018; Leonardi et al., 2024).
Table 3. PANDAS: Symptoms and Causes.
Growing evidence from basic research comes from profiling gut microbiota in children with PANS compared to control subjects. The gut microbiota of children diagnosed with PANS/PANDAS showed a marked increase in potential microbial biomarkers. These PANS/PANDAS children also showed increased activation of pathways that modulate antibody response within the gut (Quagliariello et al., 2018). Practically speaking, a comprehensive medical work-up and a complete family history of the patient diagnosed with PANS/PANDAS are in the patient’s best interest.
Screening and Assessment
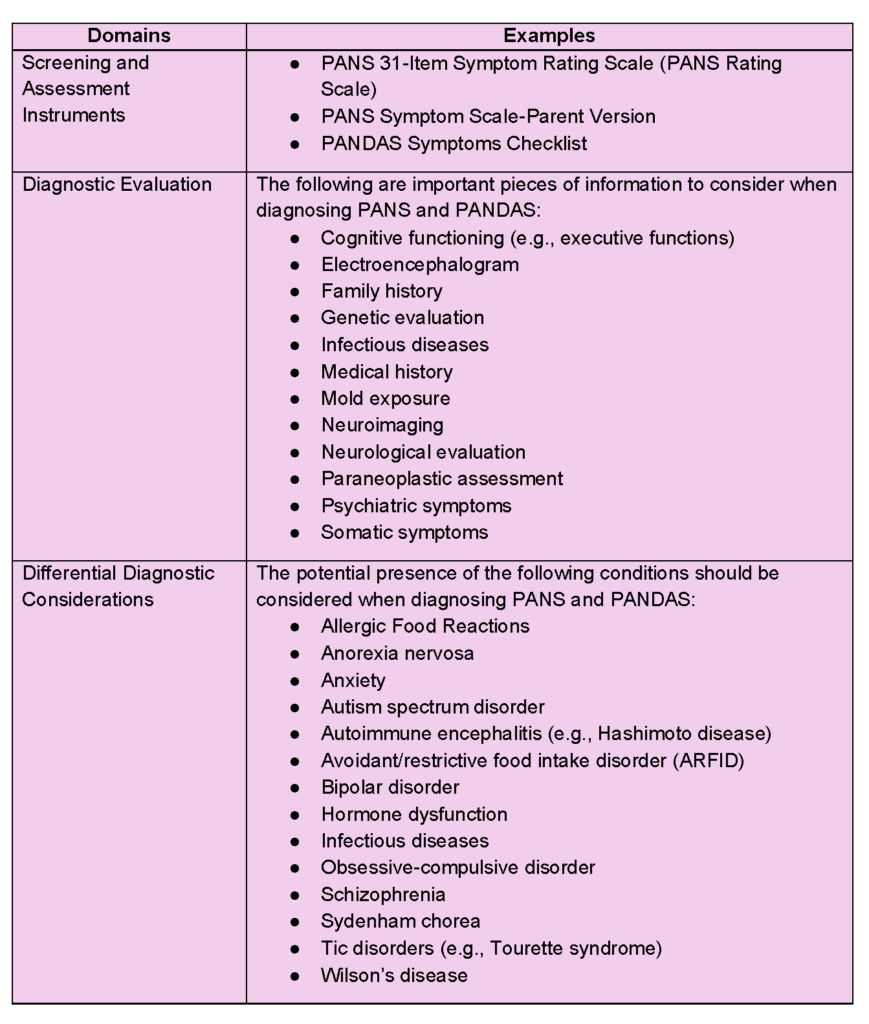
While only qualified medical doctors can formally diagnose PANS and PANDAS, mental health professionals can play a critical role in identifying psychological symptoms and associated behaviors during their screening and assessment process. Table 4 provides an overview of assessment options, essential components of a diagnostic evaluation for PANS or PANDAS, and a review of other conditions to consider during differential diagnosis (Antoine, 2024; Arcilla & Singla, 2024; Chang et al., 2015). As highlighted in this table, diagnosing PANS and PANDAS relies on information from several domains rather than a single test (PANDAS Network, 2025), including input from multiple medical and psychiatric disciplines. Ruling out other possible conditions (e.g., autoimmune conditions, psychiatric disorders, genetic disorders) is a critical step in diagnosing PANS and PANDAS (Arcilla & Singla, 2024). Consequences for the missed or incorrect diagnoses of PANS and PANDAS include ineffective interventions, persistence of symptoms, and diminished quality of life (Thienemann et al., 2017). All professionals are strongly encouraged to perform a comprehensive safety assessment (e.g., eating habits, aggression, and self-harm) as a part of the diagnostic process (Heavey & Peterson, 2019; Thienemann et al., 2017).
Table 4. Multidisciplinary Screening and Assessment of PANS and PANDAS.
Treatment and intervention
Although there is no “gold standard” (Dailey et al., 2025), treatment approaches for PANS and PANDAS typically focus on addressing (a) infections or inflammation (e.g., antibiotics), (b) psychiatric symptoms, and (c) behavioral problems (O’Hara, 2022; Sigra et al., 2018). The approach for each client should be tailored based on the presence and severity of specific symptoms (Ringer et al., 2023). A treatment approach should be comprehensive and multidisciplinary and include (a) treatment with psychoactive medications, psychotherapies, and supportive interventions, (b) addition of antimicrobial interventions to remove the source of inflammation, and (c) treatment with immunomodulatory and/or anti-inflammatory therapies that treat the disorders of the immune system (Swedo et al., 2017).
Further, a recommendation from the 2013 PANS Consensus Conference is for PANS patients or patients with only autoimmune features to undergo an immunodeficiency assessment; especially if there is evidence of a history of repeated infections, infection with an atypical organism, any genetic connection to a fatal infection of a relative, or if the medical provider is considering intravenous immunoglobulin (IVIG) treatment (Chang et al., 2015). The literature indicates that an immunodeficiency assessment should include a workup on the lymphocyte subsets (T, B, and NK-cells), quantitative assessment of immunoglobulins (IgG, IgA, IgM, IgE, and a vaccine response test for Pneumococcus and tetanus antibody titers (Chang et al, 2015; LeFon & Nahm, 2018). This immunodeficiency assessment should be performed in conjunction with continued clinical evaluation of the patient (Chang et al., 2015).
Multidisciplinary teams may be the most effective path forward in treating PANS and PANDAS, and could benefit from including infectious disease specialists, immunologists, neuropsychiatrists, nutritionists, pediatricians, and rheumatologists (Dailey et al., 2025). A critical opportunity in the treatment of PANS and PANDAS is partnering with the child’s parents and other family members (Ringer et al., 2023). These individuals often play a strong role in seeking treatment for the child and can be instrumental in helping maximize the child’s recovery (Aspire, 2025). Professionals should provide family members with educational resources that enhance their understanding of PANS and PANDAS, help establish support systems, and facilitate their participation in treatment and intervention processes (Aspire, 2025). Further, parents and other family members may benefit from brief interventions focused on coping with and accepting PANS and PANDAS, as the experience can be stressful and traumatic (Ellerkamp et al., 2023; Thienemann et al., 2017). Fortunately, many children do experience complete or near-complete recovery from PANS and PANDAS, especially with early recognition, proper treatment, and a multidisciplinary approach. According to the PANS Research Consortium, the majority of patients improve significantly or recover fully with appropriate interventions. Some may require ongoing support to manage residual symptoms or prevent symptom recurrence (Chang et al., 2015).
Table 5 provides an overview of the multidisciplinary treatment and intervention approaches, targets, and practices that professionals should consider when addressing PANS and PANDAS (Brown et al., 2017; Frankovich et al., 2017; Guido et al., 2019; Khimani et al., 2024; O’Hara, 2022; Rea et al., 2021; Swedo et al., 2015; Thienemann et al., 2016; Thienemann et al., 2017).
Table 5. Multidisciplinary Treatment and Interventions for PANS and PANDAS.

PANS/PANDAS case study
The fictitious clinical case study of Jane, a 9-year-old girl, highlights the processes and challenges involved in assessing and treating PANS/PANDAS.
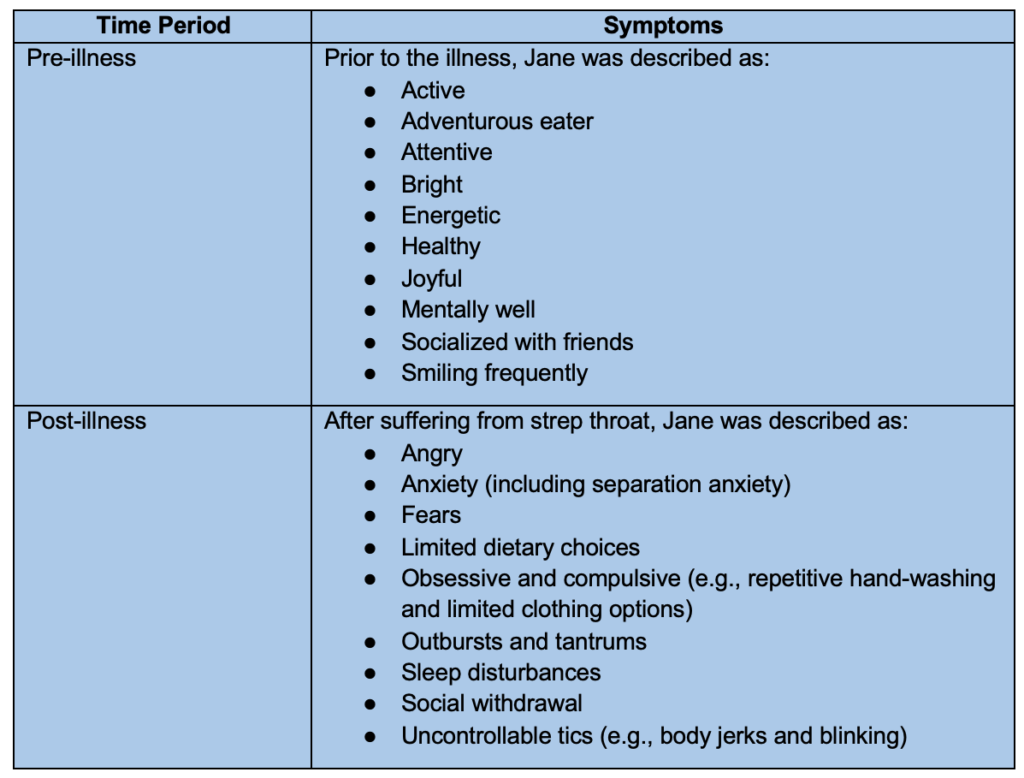
Jane’s parents sought care for their daughter at a local community behavioral health clinic after observing sudden changes in her moods (from joyful to irritable) and concerning behaviors. During this initial visit, Ms. Smith, a licensed mental health professional, conducted a screening interview along with a comprehensive biopsychosocial intake. Jane’s parents linked these sudden changes to a case of streptococcal pharyngitis (strep throat). Table 6 contrasts the differences in Jane’s mental health and behaviors before and after this illness. Despite visits to several different doctors, her parents struggled to find answers regarding their daughter’s condition. Some of the initial diagnoses included obsessive-compulsive disorder and anxiety disorder. After ruling out trauma and abnormal blood test results, Ms. Smith began to suspect PANDAS. A follow-up appointment was scheduled with Jane’s pediatrician, during which Ms. Smith shared the clinical summary she had developed. Ms. Smith adopted a biopsychosocial approach (for an overview, see Table 7; Dalgleish et al., 2020) to the development of Jane’s treatment plan and engaged other medical and allied health professionals with relevant experience.
Table 6. Jane’s Symptoms Before and After Strep Throat.
This case study highlights how mental health providers can be instrumental in identifying PANS/PANDAS, facilitating appropriate referrals, and monitoring the progress of interventions.
Integrated biopsychosocial interventions in PANS/PANDAS treatment
The complexity of Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) and Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) calls for a coordinated, multidisciplinary response grounded in the biopsychosocial model. Rather than focusing solely on immunological or neurological symptoms, recent literature underscores the value of integrating physical health, emotional development, family systems, and environmental context into treatment planning.
At the behavioral level, structured exercise and regulation of screen time have been associated with improved executive function and decreased emotional volatility in pediatric neuroimmune populations (Brown et al., 2022). Early intervention to reduce harmful behaviors—such as substance use in older children or adolescents—can also prevent compounding neurological damage and improve treatment engagement. Cognitively, interventions should target distorted thought patterns and deficits in adaptive functioning. Modified cognitive-behavioral strategies show promise in enhancing distress tolerance and cognitive flexibility in youth experiencing neuroinflammatory symptoms (Storch et al., 2023).
Nutritional rehabilitation is a vital adjunct to treatment, particularly when micronutrient deficiencies contribute to neurological or immunological dysregulation. For instance, deficiencies in vitamin D, B12, and iron have been linked to fatigue, mood dysregulation, and immune vulnerability (Klein et al., 2023). In addition, research indicates that deficiencies in B vitamins (B1, B2, B3, B5, B6, B8, B9, and B12), along with vitamin C, iron, magnesium, and zinc, may contribute to cognitive and psychological impairments, including mental and physical fatigue (Tardy et al.). Addressing these issues holistically requires collaboration between dietitians/nutritionists, pediatricians, and mental health clinicians.
Environmental adversity also plays a crucial role in symptom expression and recovery. Chronic stress, discrimination, poverty, and exposure to systemic oppression may heighten neuroinflammatory processes and impact access to care. Nguyen and Jackson (2024) describe a framework in which resilience-building, community resources, and trauma-informed practices reduce toxic stress and improve long-term outcomes in neurodiverse and immunocompromised youth.
Physiologically, successful treatment often depends on addressing sleep disturbances, gut-brain axis disruption, and underlying infections or autoimmune triggers. Frankovich et al. (2022) highlight the utility of integrated medical and psychiatric care, including immunomodulatory therapies such as IVIG or corticosteroids, to reduce flare-ups and stabilize functioning. Importantly, these interventions are more effective when paired with psychosocial support.
On the psychological front, fostering emotional intelligence, working through grief, and addressing personality traits that may compound symptom expression are essential. Exposure to adverse childhood experiences (ACEs) is not uncommon in this population and can significantly complicate diagnosis and treatment. Evidence-based trauma therapy must be integrated into care when relevant (Sullivan et al., 2023).
Finally, social dimensions—including attachment style, peer relationships, and family support—serve as both risk and protective factors. Programs that support caregiver education, enhance family cohesion, and reduce isolation for the child have demonstrated efficacy in improving treatment adherence and emotional regulation (Martinez & Lowenstein, 2023).
Taken together, a biopsychosocial approach—rather than a siloed biomedical model—offers a more comprehensive and effective pathway for supporting children diagnosed with PANS/PANDAS.
Table 7. Overview of Treatment Targets within a Multidisciplinary Biopsychosocial Approach.
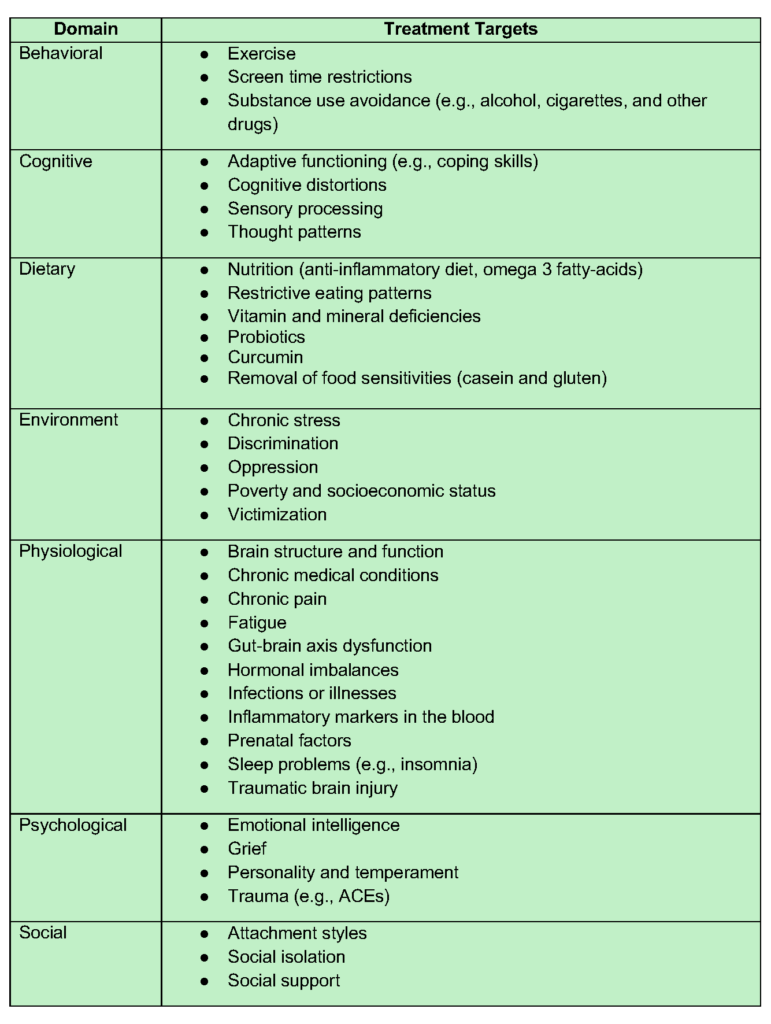
Conclusion
PANS and PANDAS, although relatively rare, are often underdiagnosed conditions in children that can respond well to timely and effective interventions. Although mental health professionals cannot diagnose PANS and PANDAS, they play a vital role in recognizing potential symptoms, initiating timely referrals, and supporting clients and families throughout the treatment process. By fostering strong collaboration with medical specialists, clinicians help ensure that children receive accurate diagnoses and comprehensive care.
Ultimately, increasing awareness of these conditions among mental health and allied professionals enhances early identification, reduces unnecessary suffering, and promotes better long-term outcomes for affected children and their families.
Key takeaways
Overview: PANS/PANDAS
- Pediatric Acute-Onset Neuropsychiatric Syndrome (PANS) and Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS) are relatively rare disorders but have significant symptoms and consequences for children and families.
- PANS and PANDAS are usually identified in children ages 4 to 13.
- Commonly shared symptoms include obsessions-compulsions, anxiety, concentration issues, fatigue, fear, memory problems, panic, sleep disturbances, and tics.
- Mental health clinicians can play an important role within a multidisciplinary team, aiding in assessment and treatment.
Symptoms and causes
- In PANDAS, Group A beta-hemolytic streptococcal (GABHS) oropharyngeal infections are implicated.
- In PANS, there is causal evidence related to pathogens, infections, or autoimmune diseases, with genetic and environmental contributions.
- PANS/PANDAS are both characterized by a rapid onset of symptoms.
- Physical symptoms may include attention control deficits, catatonia, hyperactivity, joint pain, physical movement dysfunction, refusal to eat regularly, sensory processing sensitivities, sleep problems, and motor/vocal tics.
- Psychological symptoms associated with PANS/PANDAS include aggression, anxiety, depression, developmental regression, emotion dysregulation, fear, hallucinations, impulsivity, irritability, obsessions and compulsions, panic attacks, and suicidal ideation.
Multidisciplinary screening and assessment
- Screening and assessment protocols should incorporate a multidisciplinary team of providers.
- Mental health professionals can play a critical role in identifying psychological symptoms and associated behaviors during their screening and assessment process. At the same time, only qualified medical doctors can formally diagnose PANS and PANDAS.
- There are multiple screening tools to help providers identify symptoms, including the PANS 31-Item Symptom Rating Scale (PANS Rating Scale), the PANS Symptom Scale-Parent Version, and the PANDAS Symptoms Checklist.
Multidisciplinary treatment and interventions
- Treatment for PANS and PANDAS typically focuses on addressing (a) infections or inflammation, (b) psychiatric symptoms, and (c) behavioral problems.
- Treatment is best delivered through a multidisciplinary team, involving medical, mental health, and allied providers qualified in specific areas.
Future indications
- There is a significant need for PANS/PANDAS training for mental health professionals.
- Research should continue to study psychological interventions for PANS/PANDAS.
- Clinical studies are needed to inform research on inflammatory biomarkers that can be used for diagnosis. Published data on inflammatory biomarkers in PANS patients are scarce.
- Education for parents/child advocates and clinicians regarding the sudden onset of symptoms after an infection.

Jerrod Brown, PhD, MA, MS, MS, MS, MS, MS, is a professor, trainer, researcher, and consultant with multiple years of experience teaching collegiate courses. Jerrod has completed six separate master’s degree programs and holds graduate certificates in Medical Biochemistry, Exercise Prescription, Neuroscience and the Law, Neuropsychology, Dyslexia, Autism Spectrum Disorder (ASD), Other Health Disabilities (OHD), and Traumatic Brain Injury (TBI). Currently, Jerrod is pursuing his fifth master’s degree in Applied Clinical Nutrition from Northeast College of Health Sciences.

Jeremiah Schimp, PhD, MA, LPCC, is an Assistant Professor of Psychology at Concordia University, St. Paul. He has taught psychology since 2016, specializing in counseling and psychopathology. He has worked in the mental health field since 2004, focusing his clinical career on adults diagnosed with severe and persistent mental illnesses and substance use disorders. Dr. Schimp holds a BA in Psychology, an MA in Counseling Psychology, and a PhD in Psychology with a specialization in health psychology. He is a Licensed Professional Clinical Counselor and board approved supervisor in the state of Minnesota; and holds certifications in integrative mental health, addiction, and trauma.

Shelley Mydra, DMFT, LMFT, is an Adjunct Professor of Psychology at Pepperdine University Graduate School of Education and Psychology (GSEP). She holds a Doctorate and Licensure in Marriage and Family Therapy and has extensive clinical experience. In addition to being an AAMFT Professional member holding the Approved Supervisor and Clinical Fellow designations, she is certified in Lifestyle Medicine and Pain Neuroscience Education. She serves on the advisory boards of the GSEP Board of Visitors and The Impact Collaborative.

Jenenne R. Valentino-Bottaro, PhD, LMHC-S, is a Licensed Mental Health Counselor practicing in Florida and Utah. Jenenne is an instructor at Walden University, is a dancer, and practices martial arts. In addition, Jenenne is a Certified Yoga Instructor specializing in prenatal yoga and yoga for the nervous system, Certified Bodyways Specialist and is trained in Dance for PD (Parkinson’s disease). Jenenne is the founder and owner of Vai’datha, a center committed to healing through community, connection, and expression, through the arts-in-health and arts-in-community frameworks. Beyond her private practice, Jenenne offers consultation services to community agencies, organizations, and mental health and medical practices on integrating traditional therapeutic methodologies with therapeutic dance, drumming, yoga, human-animal welfare interconnectedness practices and other expressive arts practices for chronic medical conditions, complex trauma, substance use recovery, movement disorders, PANDA/PANS and Autism Spectrum Disorder (ASD), particularly among women and girls.

Leanne Skehan DCN, MSACN, MPH, MBA, holds a Doctor of Clinical Nutrition degree from Maryland University of Integrative Health. She is a full-time faculty member at Southern New Hampshire University in the Master of Public Health program and an adjunct instructor at Northeast College of Health Sciences in the Master of Applied Clinical Nutrition program.

Bettye Sue Hennington, PhD, is a professor at the University of Mississippi Medical Center, with 24 years of teaching experience in institutions of higher learning. Her research interest is in the field of metabolomics specifically in the causes of low birth weight in newborns.

Kristy Donaldson, PhD, LPC-S, RPT-S, CHST, is a licensed professional counselor supervisor (LPC-S), registered play therapist supervisor (RPT-S™), and court-qualified expert specializing in mental health, forensic consultation, and clinical supervision. She serves as an adjunct professor at Tarleton State University, where she teaches graduate counseling courses and mentors future clinicians. With certifications in EMDR, the Enneagram, and animal-assisted counseling, Dr. Donaldson integrates research-based practices into therapy, education, and courtroom testimony.

Jennifer Sweeton, JD, PsyD, MS, MA, is a clinical and forensic psychologist specializing in the assessment and treatment of trauma- and stressor-related disorders. Dr. Sweeton owns a private practice, and is an adjunct faculty member at the University of Kansas School of Medicine. She completed her doctoral training at the Stanford School of Medicine, Palo Alto University, and the National Center for PTSD.
American Association for Marriage and Family Therapy [AAMFT]. (2026). AAMFT Code of Ethics (effective January 1, 2026). https://www.aamft.org/AAMFT/Legal_Ethics/Code_of_Ethics.aspx
American Medical Association [AMA]. (2025, June 12). AI scribes save 15,000 hours—and restore the human side of medicine. AMA News Wire. https://www.ama-assn.org/practice-management/digital-health/ai-scribes-save-15000-hours-and-restore-human-side-medicine
Asgari, E., Montaña-Brown, N., Dubois, M., Pimenta, D., & others. (2025). A framework to assess clinical safety and hallucination rates of LLMs for medical text summarisation. npj Digital Medicine, 8, 274. https://doi.org/10.1038/s41746-025-01670-7
Bateson, G. (1972). Steps to an ecology of mind: Collected essays in anthropology, psychiatry, evolution, and epistemology. Chandler Publishing.
Lockhart, E. N. S. (2025a). Jay Haley’s model of strategic family therapy: An epistemological inquiry. Philosophy, Psychiatry & Psychology, 32(4), 407–434. https://doi.org/10.1353/ppp.2025.a978088
Lockhart, E. N. S. (2025b). Mentorship and clinical supervision through Haley’s strategic model: A composite case study in legal literacy. Journal of Systemic Therapies, 43(3), 1–19. https://doi.org/10.1521/jsyt.2024.43.3.01
Lukac, P. J., Turner, W., Vangala, S., Chin, A. T., Khalili, J., Shih, Y. C. T., … & Mafi, J. N. (2025). Ambient AI scribes in clinical practice: a randomized trial. The New England Journal of Medicine AI, 2(12), AIoa2501000. https://doi.org/10.1056/AIoa2501000
Martin, J. L., & Wright, K. E. (2023). Bias in automatic speech recognition: The case of African American language. Applied Linguistics, 44(4), 613–630. https://doi.org/10.1093/applin/amac066
Mengesha, Z., Heldreth, C., Lahav, M., Sublewski, J., & Tuennerman, E. (2021). “I don’t think these devices are very culturally sensitive.”—Impact of automated speech recognition errors on African Americans. Frontiers in Artificial Intelligence, 4, 725911. https://doi.org/10.3389/frai.2021.725911
Mess, S. A., Mackey, A. J., & Yarowsky, D. E. (2025). Artificial intelligence scribe and large language model technology in healthcare documentation: Advantages, limitations, and recommendations. Plastic and Reconstructive Surgery Global Open, 13(1), e6450. https://doi.org/10.1097/GOX.0000000000006450
Tierney, A. A., Gayre, G., Hoberman, B., Mattern, B., Ballesca, M., Wilson Hannay, S. B., … & Lee, K. (2025). Ambient artificial intelligence scribes: Learnings after 1 year and over 2.5 million uses. The New England Journal of Medicine Catalyst Innovations in Care Delivery, 6(5), https://doi.org/10.1056/CAT.25.0040
Topaz, M., Peltonen, L. M., & Zhang, Z. (2025). Beyond human ears: Navigating the uncharted risks of AI scribes in clinical practice. npj Digital Medicine, 8(1), 569. https://doi.org/10.1038/s41746-025-01895-6
Other articles
When the Chart Is Watching Back: AI, Consent, and Control in Teletherapy Documentation
Picture this: you finish a teletherapy session, rich with layered family dynamics, unspoken tensions, and a breakthrough moment in relational co-construction, and open the electronic health record to chart. But the platform has already been at work. An AI-generated summary sits there, neatly formatted, highlighting symptoms, diagnoses, and suggested next steps. It asks for your review, edits, approval; it even asks you to rate how well it did.
Ezra N. S. Lockhart, PhD
How Can Marriage and Family Therapists Help Racially and Ethnically Minoritized Individuals Navigating Online Dating?
For many, dating app platforms represent hope and possibility. For racially and ethnically minoritized individuals, however, the experience is often far more complicated.
Eman Tadros, PhD, Annemarie Sohn, MA, & Jixuan Zhao, MSW
AI in the Therapy Room: A Client’s Need for Informed Consent
Recently, an acquaintance of an acquaintance (let’s call her Dina) heard that I was a therapist and an educator and asked if she could chat with me (this write-up was approved by her). She shared that she discovered her therapist was using AI to partially conduct their sessions.
Mudita Rastogi, PhD